How do we distinguish enzymes obeying a sequential or concerted reaction mechanism?
'From idea to published article': in this series (introduced here), authors highlight one of their recent papers and the story of the work behind it.
Featured article:
Morea, V., Angelucci, F., Tame, J.R.H., Di Cera, E. and Bellelli, A. (2022) Structural basis of sequential and concerted cooperativity. Biomolecules 12, 1651. https://doi.org/10.3390/biom12111651
The most influential models that can explain cooperativity and heterotropic regulation of enzymes, receptors, and in general of ligand-binding proteins are those developed in the 1960s by Monod, Wyman and Changeux [1] (MWC – the concerted model), and by Koshland, Neméthy and Filmer [2] (KNF – the sequential model). These models do make structural predictions, but a systematic assessment on the correspondence between prediction and experimental evidence is incomplete at best; moreover, assessment of the reaction mechanism has been in many cases quite uncertain, given that both models may be able to describe the functional behaviour of the majority of cooperative proteins and enzymes. The differences between the two mechanisms are listed in Table 1 of our recent paper [3].
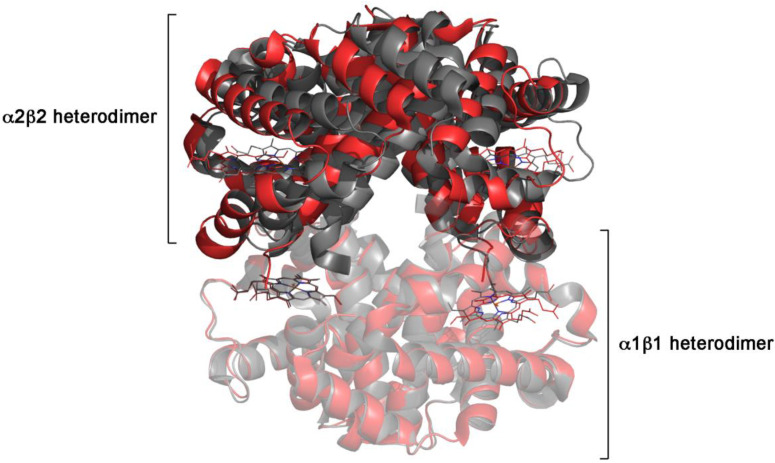
I discussed this problem with many colleagues over several years, and finally we could collect a group having the necessary types of expertise: two crystallographers (JRHT and FA), one structural bioinformatician (VM) and an expert in protein thermodynamics (EDC). We were ready! Formalizing the question our future paper aimed to answer required many further discussions and more beer than ink, but, in the end, we agreed on this scope: to look for ligation-dependent structure parameters able to distinguish the MWC from the KNF reaction mechanism. We started by looking for ligand-binding proteins whose reaction mechanism is known with certainty. This step was less easy than it may seem, because in many cases the reaction mechanism is presumed, without obvious proof. We considered proof of a concerted, MWC-like mechanism the symmetry of incompletely liganded states (whose structure unfortunately is available only in very few cases), or the demonstration of a ligand-independent structure change. Since the concerted model is not compatible with negative homotropic cooperativity, we selected proteins having this behaviour as examples of the sequential, KNF-like mechanism (see the proteins listed in Table 2 of Morea et al. [3]).
Once these problems were solved, the remaining work was straightforward: VM determined the extent of the ligand-induced structure changes in the tertiary and quaternary structures, and we obtained a neat correlation with function: in MWC-like proteins the tertiary structure change (estimated from the RMSD) accounts for approx. 70–75% of the overall structure change; in KNF-like proteins the ratio is inverted and the tertiary structure change accounts for 60–80% of the overall change. In retrospect, we were quite lucky: first, because our heterogeneous group worked quite well together; second, because we found a clear-cut structural feature whose correlation with the reaction mechanism appears solid and consistent with the expectations, in spite of, or perhaps thanks to, the relatively small number of proteins studied; and third, because during this study we found matter for further investigation on the structure-function relationship of allosteric proteins, which I shall describe in a future post.
References
1. Monod, J., Wyman J. and Changeux J.P. (1965) On the nature of allosteric transitions: a plausible model. J. Mol. Biol. 8, 88–118. https://doi.org/10.1016/s0022-2836(65)80285-6
2. Koshland, D.E., Jr, Némethy, G. and Filmer, D. (1966) Comparison of experimental binding data and theoretical models in proteins containing subunits. Biochemistry 5, 365–385. https://doi.org/10.1021/bi00865a047
3. Morea, V., Angelucci, F., Tame, J.R.H., Di Cera, E. and Bellelli, A. (2022) Structural basis of sequential and concerted cooperativity. Biomolecules 12, 1651. https://doi.org/10.3390/biom12111651




Join the FEBS Network today
Joining the FEBS Network’s molecular life sciences community enables you to access special content on the site, present your profile, 'follow' contributors, 'comment' on and 'like' content, post your own content, and set up a tailored email digest for updates.