Mathias Uhlen: "A large part of the cutting-edge research in biology and medicine is technology..."

What do you see as the most important developments in your field in the past 15 years?
Firstly, the recent clinical success of biological drugs, which now constitute the majority of the best-selling drugs in the world. Secondly, the development of high-throughput methods for analysing biological samples, such as next-generation sequencing, proteomics, tissue microarrays and imaging technologies. And thirdly, the development of systems biology to analyse large amounts of quantitative biological data.
What drew you to your research field?
In the beginning of my career, I was focusing my research on molecular biology and DNA sequencing. I was involved in large-scale sequencing projects, but also technology development, such as the development in our group of the Pyrosequencing method leading to the first next-generation instruments (454/Roche). When the first human genome was published in early 2000, I decided to use the knowledge from the genome sequence to try to map the corresponding proteome using immune-based methods combined with other omics technologies. A generous grant from a non-profit foundation allowed me to set up a large consortium of researchers combining protein science with genomics, image analysis, pathology and systems biology. We have now carried out this massive proteome mapping effort for the last 15 years and published more than 500 peer-reviewed articles and also launched an open access database for researchers both in academia and industry.
Tell us about a favourite published paper from your lab
During the last five years, we have published a series of papers within the framework of the Human Protein Atlas (www.proteinatlas.org) focusing on particular aspects of the genome-wide analysis of the human proteins: the Tissue Atlas showing the distribution of the proteins across all major tissues and organs in the human body (Uhlen, M. et al. Science 2015), the Cell Atlas showing the subcellular localization of proteins in single cells (Thul, P.J. et al. Science 2017) and the Pathology Atlas showing the impact of protein levels for survival of patients with cancer (Uhlen, M. et al. Science 2017). Obviously, to be able to contribute to the proteome-wide mapping of all human proteins for the first time in scientific history is extremely exciting and rewarding.
What aspects of your life as a researcher do you most enjoy?
There are several aspects of being a life science researcher that I truly enjoy. First, to have the opportunity to map human biology in a manner not possible before and to probe the magnificent molecular details of being human. Second, to plan and execute projects in the field of human health and disease and to see patients benefit from drugs developed as part of my research. I also enjoy very much the discussions of primary data from experiments with young team members to define the most important “take-home” messages. Finally, to be able to interact with bright fellow researchers both nationally and internationally and to be able to travel and see different parts of the world.
What comes first: technique or biological question?
Both. A large part of the cutting-edge research in biology and medicine is technology – and data-driven and new technologies allows the community to ask questions with higher precision and quality. It is therefore important to have research with the sole objective to improve or create new technology platforms. However, it is of course also important to have researchers that use the developed technology on questions of biological relevance.
Introduction to Mathias Uhlen's work
Research summary
Mathias Uhlen's research is focused on protein science, antibody engineering and precision medicine and ranges from basic research in human and microbial biology to more applied research, including clinical applications in cancer, infectious diseases, cardiovascular diseases, autoimmune diseases and neurobiology. Since 2003, he has led an international effort to systematically map the human proteome and transcriptome to create a Human Protein Atlas using antibodies and various omics technologies. This effort has so far resulted in the Tissue Atlas (2015) showing the distribution of proteins across human tissues and organs, the Cell Atlas (2016) showing the subcellular location of human proteins in single cells and the Pathology Atlas (2017) showing how cancer patient survival is tied to RNA and protein levels.
Lab webpage: www.proteinatlas.org
Two recent/key papers:
Uhlen, M. et al. (2015) Tissue-based map of the human proteome. Science 347,1260419, doi: 10.1126/science.1260419
Uhlen, M. et al (2017) A pathology atlas of the human cancer transcriptome. Science 357, eaan2507, doi: 10.1126/science.aan2507
More information on the FEBS Sir Hans Krebs medal and plenary lecture at the 44th FEBS Congress
The Sir Hans Krebs medal is awarded annually by FEBS for outstanding achievements in Biochemistry and Molecular Biology or related sciences: https://www.febs.org/our-activities/awards/medals/
Mathias Uhlen will be presented with the medal at the 44th FEBS Congress in Krakow, Poland on Monday 8th July where he will deliver a lecture on ‘The Human Protein Atlas – implications for human biology, drug development and precision medicine’: 2019.febscongress.org
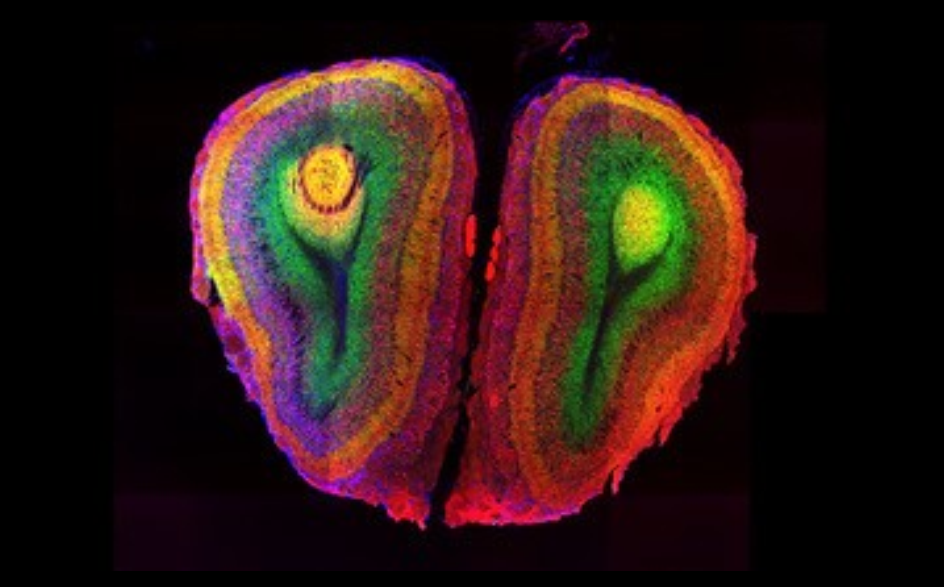
Top image of post: Photo from the Human Protein Atlas: Olfactory bulb of the mouse brain. Source: Jan Mulder, SciLIfeLab, Stockholm, Sweden.





Join the FEBS Network today
Joining the FEBS Network’s molecular life sciences community enables you to access special content on the site, present your profile, 'follow' contributors, 'comment' on and 'like' content, post your own content, and set up a tailored email digest for updates.