Celebrating the 2017 Nobel Prize in Physiology or Medicine

Circadian rhythms synchronize all biological processes with the phases of the solar day. In this FEBS Press Virtual Issue, we present a collection of articles from The FEBS Journal, FEBS Letters and FEBS Open Bio on the molecular coordinates of the circadian clock. Selected Review articles will introduce you to the molecular mechanisms of the circadian clock in mammals[i], the roles of clock proteins of the inner circadian clock, as elucidated based on structural analyses[ii], and drug targeting approaches to manipulate circadian rhythms[iii].
If you wish to delve deeper into the molecular and biochemical mechanisms involved in the regulation of the circadian clock, we invite you to take a close look at our most recent research articles on, for example, transcriptional targets of the circadian clock genes such as PER2, Cryptochrome 2[iv],[v]. Moreover, you can learn more on the roles of circadian oscillators in physiology and disease[vi],[vii],[viii],[ix] and how environmental factors, such as microgravity[x] or ketogenic diet[xi], may influence the clock. Finally, a collection of papers published back in the 1970s[xii],[xiii], 1980s[xiv] and 1990s[xv],[xvi],[xvii] and 2000s[xviii] may give you a taste of how the field has evolved during the past four decades.
We hope you enjoy our collection and come back to FEBS Press for reading or submitting further work unravelling the secrets of Chronobiology.
[i] Robinson I and Reddy AB. Molecular mechanisms of the circadian clockwork in mammals. FEBS Letters 588 (15), 2015. DOI: 10.1016/j.febslet.2015.05.024
[ii] Merbitz-Zahradnik T and Wolf E. How is the inner circadian clock controlled by interactive clock proteins? Structural analysis of clock proteins elucidates their physiological role. FEBS Letters 589 (14), 2014. DOI: 10.1016/j.febslet.2014.06.005
[iii] Wallach T and Kramer A. Chemical chronobiology: Towards drugs manipulating time. FEBS Letters 589 (14), 2015. DOI: 10.1016/j.febslet.2015.04.059
[iv] Kobayashi M et al. A circadian cock gene, PER2, activates HIF-1 as an effector molecule for recruitment of HIF-1α to promoter regions of its downstream genes. The FEBS Journal 20 Sept 2017. DOI: 10.1111/febs.14280
[v] Facella P et al. Cryptochrome 2 extensively regulates transcription of the chloroplast genome in tomato. FEBS Open Bio 7 (4), 2017. DOI: 10.1002/2211-5463.12082
[vi] Melkani GC et al. TRiC/CCT chaperonins are essential for maintaining myofibril organization, cardiac physiological rhythm, and life span. FEBS Letters 30 Sept 2017. DOI: 10.1002/1873-3468.12860
[vii] Maurer C et al. The CREB-binding protein affects the circadian regulation of behaviour. FEBS Letters 590(18), 2017. DOI: 10.1002/1873-3468.12336
[viii] Yamashita K et al. The effects of the heme precursor 5-aminolevulinic acid (ALA) on REV-ERBα activation. FEBS Open Bio 4(1), 2014. DOI: 10.1016/j.fob.2014.03.010
[ix] Nitanda Y et al. 3′–UTR-dependent regulation of mRNA turnover is critical for differential distribution patterns of cyclic gene mRNAs. The FEBS Journal 281(1), 2014. DOI: 10.1111/febs.12582
[x] Ranieri D et al. Microgravity influences circadian clock oscillation in human keratinocytes. FEBS Open Bio 5(1), 2015. DOI: 10.1016/j.fob.2015.08.012
[xi] Oishi K et al. Ketogenic diet and fasting induce the expression of cold-inducible RNA-binding protein with time-dependent hypothermia in the mouse liver. FEBS Open Bio 3(1), 2013. DOI: 10.1016/j.fob.2013.03.005
[xii] Selkov E et al. Stabilization of Energy Charge, Generation of Oscillations and Multiple Steady States in Energy Metabolism as a Result of Purely Stoichiometric Regulation. European Journal of Biochemistry 59(1), 1975. DOI: 10.1111/j.1432-1033.1975.tb02436.x
[xiii] Leong TY and Schweiger HGl. The Role of Chloroplast-Membrane-Protein Synthesis in the Circadian Clock. European Journal of Biochemistry 98(1), 1979. DOI: 10.1111/j.1432-1033.1979.tb13176.x
[xiv] Sheikh SP et al. Y1 and Y2 receptors for neuropeptide Y. FEBS Letters 245(1-2), 1989. DOI: 10.1016/0014-5793(89)80223-6
[xv] Davies SP et al. Diurnal rhythm of phosphorylation of rat liver acetyl – CoA carboxylase by the AMP-activated protein kinase, demonstrated using freeze-clamping. European Journal of Biochemistry 203(3), 1992. DOI: 10.1111/j.1432-1033.1992.tb16591.x
[xvi] Eva C et al. The murine NPY-1 receptor gene Structure and delineation of tissue-specific expression. FEBS Letters 314(3), 1992. DOI: 10.1016/0014-5793(92)81490-D
[xvii] Koike N et al. Identification of the mammalian homologues of the Drosophila timeless gene, Timeless1. FEBS Letters 441(3), 1998. DOI: 10.1016/S0014-5793(98)01597-X
[xviii] Nagel R et al. The miRNA-192/194 cluster regulates the Period gene family and the circadian clock. The FEBS Journal 276(19), 2009. DOI: 10.1111/j.1742-4658.2009.07229.x
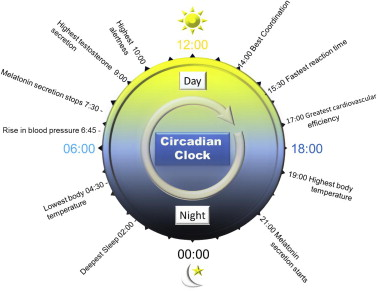
Poster image: From the Nobel Prize Press Release.





Join the FEBS Network today
Joining the FEBS Network’s molecular life sciences community enables you to access special content on the site, present your profile, 'follow' contributors, 'comment' on and 'like' content, post your own content, and set up a tailored email digest for updates.