Structural analysis of functionally diverse and structurally polymorphic amyloid assemblies
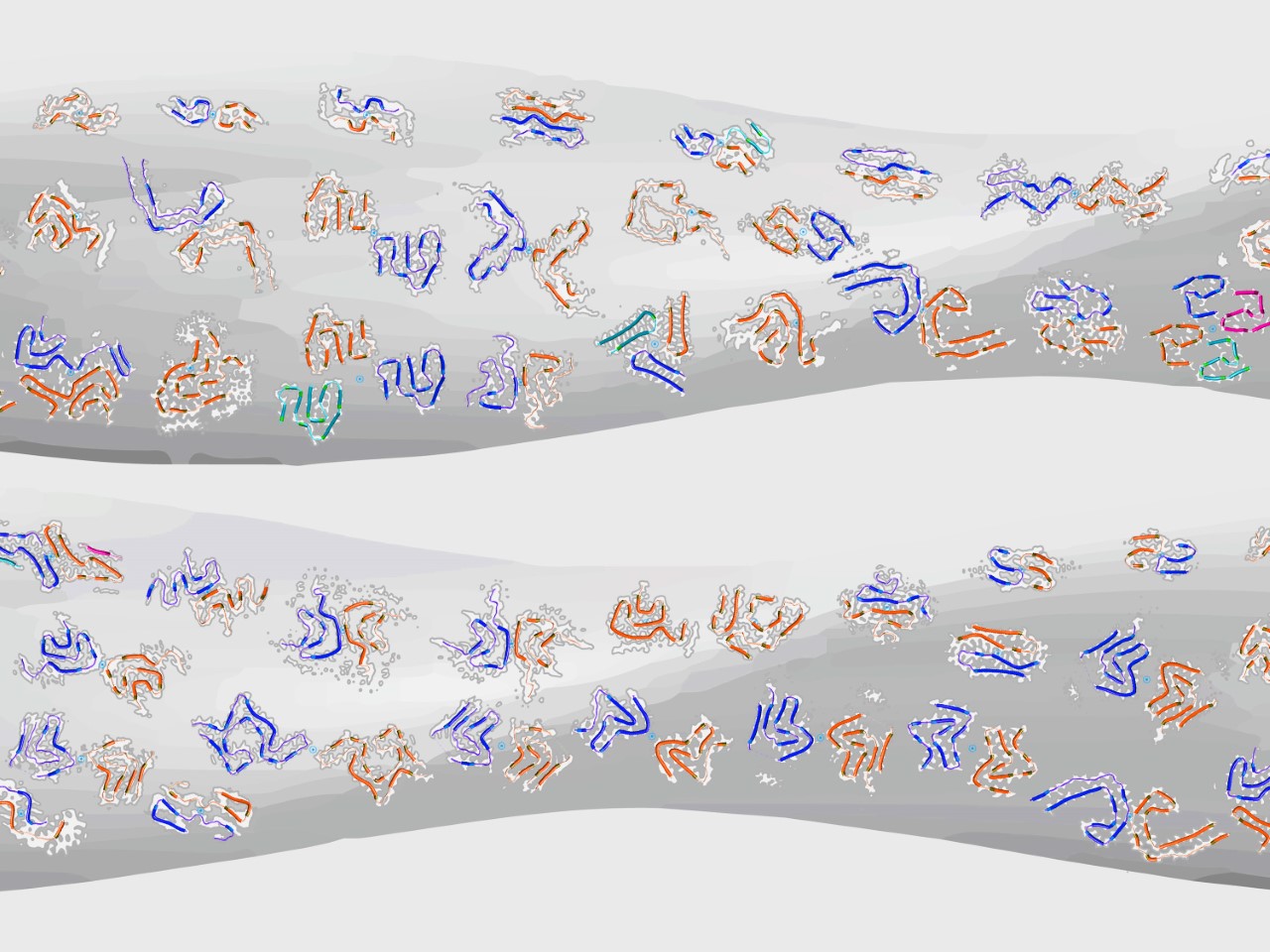
Amyloid fibrils are miniature, nanometres wide, fibrillar structures formed from proteins (reviewed in [1]). They are particularly important to understand because of their involvement in the pathology of many devastating human diseases. Well-studied pathologically linked amyloid structures include fibrils formed from amyloid-b peptides (Alzheimer's disease), tau (Alzheimer's disease and other tauopathies), a-synuclein (Parkinson's disease, multiple system atrophy and other synucleinopathies) and islet amyloid polypeptide (IAPP, also called amylin, involved in type-2 diabetes).
However, far from all amyloid structures are associated with disease. Some amyloid fibrils perform useful biological functions in a diverse range of organisms from bacteria and yeast to humans (see these recent reviews: [2-4]). Amyloid formed from curli (biofilm formation in bacteria), hydrophobins (surface modulation for growth and development in filamentous fungi), Sup35p (epigenetic switch in yeast), Ure2p (nitrogen catabolism in yeast), spidroin (spider silk component), Orb2 (memory persistence in fruit fly), pmel17 (melanin formation in humans) are examples of well-studied functional amyloid structures.
The diversity of biological effects or functions linked to amyloid structures begs the question of why some amyloid structures are linked to pathology while others are tolerated, even useful for their host organisms. Some ideas that may answer this question have started to emerge from comparing and contrasting the detailed structural models of disease-associated amyloid fibrils versus those of functional amyloid fibrils. The emerging ideas include suggestions that the fibril core hydrophobicity, the fibril surface properties, the surface interactions, and the stability of the fibrils, may be key biophysical and structural properties differentiating biologically functional amyloid structures from those associated with disease.
The diversity of amyloid structures, called polymorphs, that can be assembled even from a single type of protein building blocks is a manifestation of the biological and structural complexities associated with amyloid structures. Amyloid polymorphism has added considerable challenge to the study of structure-function relationships of amyloid (reviewed in [5]). Recent advances in the methods to determine protein structures, such as cryo-electron microscopy (cryo-EM) and solid-state nuclear magnetic resonance spectroscopy (ssNMR) have revealed that amyloid polymorphism is indeed omnipresent, at least in human disease-associated amyloid, with the characteristics of an amyloid disease being linked to very specific amyloid polymorphs. However, what decides the precise arrangements and the range of possible different polymorphs and their biological effects remains to be discovered. Therefore, understanding of the occurrence and the extent of amyloid polymorphs is essential if we are to understand their diverse roles in human disease as well as in essential biological functions.
To resolve the ‘polymorph distribution’ of potentially mixed and highly heterogeneous amyloid populations, we have recently developed an individual filament level structural analysis approach based on atomic force microscopy (AFM) image analysis [6]. Although cryo-EM and ssNMR methods have been successfully employed in recent years to determine the structures of amyloid fibrils with high resolution detail, they rely on ensemble averaging of fibril structures in the entire sample or significant subpopulations. Individual filament level structural analysis by helical 3D reconstruction of twisted amyloid fibrils benefits from the high signal-to-noise seen on AFM images and does not require averaging across multiple filaments or even segments of the same filament, allowing the polymorphic landscape of amyloid to be resolved [7]. Helical 3D reconstruction of twisted filament surface envelopes from AFM images gives access to structural parameters individual to each reconstructed amyloid fibril such as the cross-sectional shape and size, as well as the structure of the fibril surface that is accessible for potentially important biological interactions (e.g. [8]). Thus, the structural variations between individual fibrils within complex populations [9] are preserved and resolved during reconstruction. In addition, the 3D information from AFM helical reconstruction algorithm can be integrated with cryo-EM data, enabling direct quantitative comparison between experimental AFM images and the increasing numbers of cryo-EM datasets deposited in the Electron Microscopy Data Bank (EMDB), as well as allowing individual filament level identification of fibril polymorphs observed on AFM images [10]. Watch an illustrative video from this last paper.
Together, these advances show the utility of individual particle structural analysis using AFM to map the polymorphic landscape of amyloid assembly and provide a workflow of how cryo-EM data can be incorporated into AFM image analysis to facilitate an integrated structural analysis of amyloid polymorphism.
References
- Ke, P. C., R. Zhou, L. C. Serpell, R. Riek, T. P. J. Knowles, H. A. Lashuel, E. Gazit, I. W. Hamley, T. P. Davis, M. Fandrich, D. E. Otzen, M. R. Chapman, C. M. Dobson, D. S. Eisenberg, and R. Mezzenga. "Half a Century of Amyloids: Past, Present and Future." Chem Soc Rev 49, no. 15 (2020): 5473-509.
- Jackson, M. P., and E. W. Hewitt. "Why Are Functional Amyloids Non-Toxic in Humans?" Biomolecules 7, no. 4 (2017).
- Shanmugam, N., Modg Baker, S. R. Ball, M. Steain, C. L. L. Pham, and M. Sunde. "Microbial Functional Amyloids Serve Diverse Purposes for Structure, Adhesion and Defence." Biophys Rev 11, no. 3 (2019): 287-302.
- Ulamec, S. M., and S. E. Radford. "Spot the Difference: Function Versus Toxicity in Amyloid Fibrils." Trends Biochem Sci 45, no. 8 (2020): 635-36.
- Lutter, L., L. D. Aubrey, and W. F. Xue. "On the Structural Diversity and Individuality of Polymorphic Amyloid Protein Assemblies." J Mol Biol 433, no. 20 (2021): 167124.
- Lutter, L., C. J. Serpell, M. F. Tuite, L. C. Serpell, and W. F. Xue. "Three-Dimensional Reconstruction of Individual Helical Nano-Filament Structures from Atomic Force Microscopy Topographs." Biomol Concepts 11, no. 1 (2020): 102-15.
- Aubrey, L. D., B. J. F. Blakeman, L. Lutter, C. J. Serpell, M. F. Tuite, L. C. Serpell, and W. F. Xue. "Quantification of Amyloid Fibril Polymorphism by Nano-Morphometry Reveals the Individuality of Filament Assembly." Commun Chem 3 (2020): 125.
- Koloteva-Levine, N., L. D. Aubrey, R. Marchante, T. J. Purton, J. R. Hiscock, M. F. Tuite, and W. F. Xue. "Amyloid Particles Facilitate Surface-Catalyzed Cross-Seeding by Acting as Promiscuous Nanoparticles." Proc Natl Acad Sci U S A 118, no. 36 (2021).
- Konstantoulea, K., P. Guerreiro, M. Ramakers, N. Louros, L. D. Aubrey, B. Houben, E. Michiels, M. De Vleeschouwer, Y. Lampi, L. F. Ribeiro, J. de Wit, W. F. Xue, J. Schymkowitz, and F. Rousseau. "Heterotypic Amyloid Beta Interactions Facilitate Amyloid Assembly and Modify Amyloid Structure." EMBO J 41, no. 2 (2022): e108591.
- Lutter, L., Y. K. Al-Hilaly, C. J. Serpell, M. F. Tuite, C. M. Wischik, L. C. Serpell, and W. F. Xue. "Structural Identification of Individual Helical Amyloid Filaments by Integration of Cryo-Electron Microscopy-Derived Maps in Comparative Morphometric Atomic Force Microscopy Image Analysis." J Mol Biol 434, no. 7 (2022): 167466.
Top image provided by the author





Join the FEBS Network today
Joining the FEBS Network’s molecular life sciences community enables you to access special content on the site, present your profile, 'follow' contributors, 'comment' on and 'like' content, post your own content, and set up a tailored email digest for updates.