Editor's Choice Articles - December 2020

GPR120 facilitates cholesterol efflux in macrophages through activation of AMPK signaling pathway
Cholesterol efflux from macrophages is the initial step of reverse cholesterol transport, a process that protects against atherosclerosis by removing cholesterol from the arterial wall. G‐protein‐coupled receptor (GPR) 120 has a poorly defined role in macrophage foam cell formation, the hallmark of atherosclerotic plaques.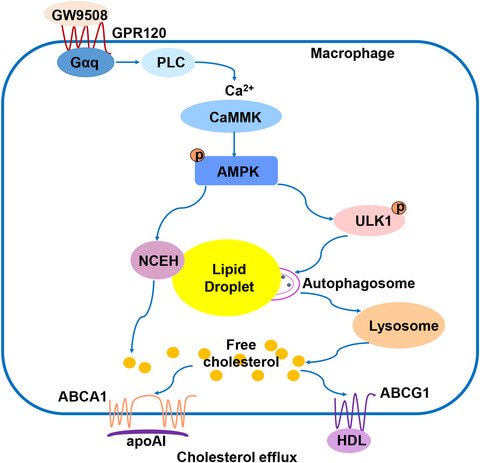 Here, Weiqing Tang and co‐authors show that stimulation of GPR120 by its agonist GW9508 leads to phosphorylation of AMPK via the PLC/Ca2+/CaMMK‐dependent pathway, upregulating the expression of neutral cholesterol ester hydrolase and ATP‐binding cassette transporters ABCA1 and ABCG1. These results show that GPR120 promotes cholesterol ester hydrolysis and ABCA1‐ and ABCG1‐ mediated cholesterol efflux from macrophage foam cells.
Here, Weiqing Tang and co‐authors show that stimulation of GPR120 by its agonist GW9508 leads to phosphorylation of AMPK via the PLC/Ca2+/CaMMK‐dependent pathway, upregulating the expression of neutral cholesterol ester hydrolase and ATP‐binding cassette transporters ABCA1 and ABCG1. These results show that GPR120 promotes cholesterol ester hydrolysis and ABCA1‐ and ABCG1‐ mediated cholesterol efflux from macrophage foam cells.
Read the Original Article here: https://febs.onlinelibrary.wiley.com/doi/10.1111/febs.15310
Vimentin S‐glutathionylation at Cys328 inhibits filament elongation and induces severing of mature filaments in vitro
Vimentin intermediate filaments are a major component of the cytoskeleton in cells of mesenchymal origin. Oxidative modification of vimentin Cys328 is known to affect the assembly of the filament. Using atomic force microscopy, electron microscopy and a novel hydrogen–deuterium exchange mass spectrometry, the authors investigated the structural impact of S‐nitrosylation and S‐glutathionylation on human vimentin oligomerisation in vitro. 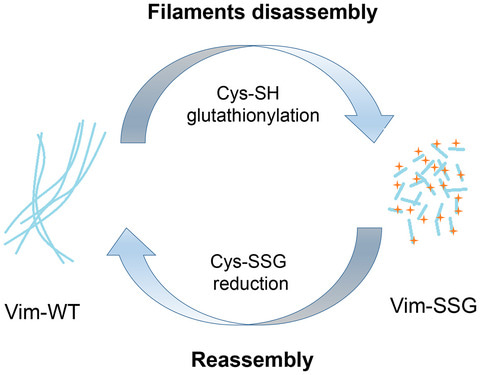 They show that S‐glutathionylation does not affect the lateral association of tetramers to unit‐length filaments, but blocks the longitudinal assembly of vimentin into long filaments and causes extensive fragmentation of preformed vimentin filaments. Thus, this modification may modulate the dynamic rearrangements of vimentin intermediate filaments in vivo.
They show that S‐glutathionylation does not affect the lateral association of tetramers to unit‐length filaments, but blocks the longitudinal assembly of vimentin into long filaments and causes extensive fragmentation of preformed vimentin filaments. Thus, this modification may modulate the dynamic rearrangements of vimentin intermediate filaments in vivo.
Read the Original Article here: https://febs.onlinelibrary.wiley.com/doi/10.1111/febs.15321





Join the FEBS Network today
Joining the FEBS Network’s molecular life sciences community enables you to access special content on the site, present your profile, 'follow' contributors, 'comment' on and 'like' content, post your own content, and set up a tailored email digest for updates.