Highlights from FEBS Letters _November 2017
hSnd2 protein represents an alternative targeting factor to the endoplasmic reticulum in human cells
Lang S. and colleagues, FEBS Letters
DOI: 10.1002/1873-3468.12831
The SRP (signal recognition particle) and GET (guided entry of tail-anchored proteins) pathways target proteins to the endoplasmic reticulum (ER). Another ER-targeting pathway, SRP-independent (SND), has been identified in yeast. Here, the authors present the first functional characterization of a human SND pathway component, hSND2. An ER membrane protein, hSnd2 preferentially targets proteins with a C-terminal transmembrane domain. The study provides insights into distinct and overlapping functions of the ER-targeting routes.
Exploiting off‐targeting in guide‐RNAs for CRISPR systems for simultaneous editing of multiple genes
Nielsen J. and colleagues, FEBS Letters
In CRISPR genome editing the Cas9 nuclease is directed to select genome sites by a single guide-RNA (gRNA). The design of promiscuous gRNAs might be useful, for example in cases where multiple genes should be edited. Thus, Ferreira et al. developed an algorithm to design such promiscuous gRNAs that may recognize the region of interest in a defined list of genes in any genome.
Dissecting the structure–function relationship in lysozyme domain of mycobacteriophage D29‐encoded peptidoglycan hydrolase
Jain V. and colleagues, FEBS Letters
Bacteriophage lysins degrade bacterial cell wall peptidoglycans during virion release. Here, Joshi et al. characterize the central lysozyme domain of Mycobacteriophage D29 Lysin A, which can hydrolyze peptidoglycans of both Gram-positive and Gram-negative bacteria. The authors map the active site and identify critical residues conferring stability and functionality. The results might enable the development of mycobacteriophage-based broad-spectrum antibacterials.
TRiC/CCT chaperonins are essential for maintaining myofibril organization, cardiac physiological rhythm, and lifespan
Bernstein, S. and colleagues, FEBS Letters
The TRiC/CCT chaperonins form a protein folding complex whose clients include the cardiac contractile proteins. This study shows that knockdown of TRiC subunits in Drosophila cardiomyocytes results in cardiac dysfunction and shortened lifespan. Disruption of circadian rhythm exacerbates the cardiac dysfunction in TRiC hypomorphs, thus revealing an intriguing link between circadian rhythm, TRiC activity, and cardiac function.
The CDK inhibitor p21 is a novel target gene of ATF4 and contributes to cell survival under ER stress
Hayashi, H. and colleagues, FEBS Letters
The activating transcription factor 4 (ATF4) determines cell fate in response to endoplasmic reticulum (ER) stress by activating pro-survival signals or, under severe ER stress, by inducing apoptosis. Here, the authors identify the cell cycle inhibitor p21 as a transcriptional target of ATF4 that is upregulated upon ER stress. Knockdown of p21 increases ER stress-induced apoptosis, indicating that p21 is a pro-survival mediator of ATF4.
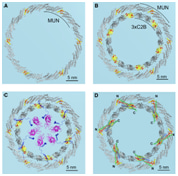
Hypothesis – buttressed rings assemble, clamp, and release SNAREpins for synaptic transmission
Rothman J.E. et al, FEBS Letters
How can neurotransmitters be synchronously released from synaptic vesicles (SVs) within milliseconds? The 'buttressed rings' model supports the assembly and clamping of SNAREpins — the membrane-fusion driving complexes forming between SVs and the plasma membrane — as well as SV release in response to action-potential triggered Ca2+ entry. The MUN and C2B domains of the SNAREpin components Munc13 and Synaptotagmin (Syt) may assemble in two concentric and co-planar rings: an outer MUN domain-based hexameric ring and an inner ring of 18 Syt C2B domains that control reactions by changing orientation on and between membranes.




Join the FEBS Network today
Joining the FEBS Network’s molecular life sciences community enables you to access special content on the site, present your profile, 'follow' contributors, 'comment' on and 'like' content, post your own content, and set up a tailored email digest for updates.