PCNA: The hidden simplicity which rules our intricate lives
What is life? All of us might have asked ourselves this question without finding a suitable answer that covered the whole thing. Do you see? How can such a simple question be as difficult to answer? This constant antithesis that we find in our complex lives is what fascinates me most. Anyway, going back to the question, innumerable correct answers can arise depending on the perspective of those who contemplate the question; some could explain that our life is the result of a divine force, others would say that our life is nothing more than the meaning that we want to give it, and others would reply that life is a group of complex biological processes.
According to the vast majority of scientists, to define an organism as a living being, it must accomplish some basic characteristics, such as its ability to reproduce, grow, metabolize, and be responsive to the environment [1]. Therefore, we consider a single yeast cell as alive as a pluricellular organism like a human being, but how can a baker’s yeast cell share common features with us? The budding yeast Saccharomyces cerevisiae (S. cerevisiae) is an eukaryotic single-celled microorganism that shares around 30% of its genome with us and, in addition, 20% of human disease genes have counterparts in yeast [2]. Even though its genome is smaller and more compact than the human genome, the way these cells replicate their entire genome before dividing into two daughter cells is surprisingly conserved among eukaryotes. This conservation allows us to unveil some of the most fundamental and intricate biological processes that are sometimes harder or even impossible to study in human cells. For this reason, my current research is focused on uncovering the regulatory mechanisms of the eukaryotic DNA replication using S. cerevisiae as a biological model. Since error accumulation during DNA replication can lead to malignant transformation in human cells, a full understanding of this vital process is key to tackling crucial questions such as cancer origin and emerging therapy resistances.
DNA replication is a fundamental biological process essential for living organisms, ensuring faithful genome duplication to propagate genetic material across generations. To this end, several proteins must form complex molecular machines that will need to function in the right place and at the exact time to ensure a timely and reliable DNA duplication. Then, who or what determines where and when each protein has to perform its enzymatic activity? How is it possible that almost invisible and non-intelligent things know how to act perfectly to develop such a complex process coordinately? The answers to these questions are still not fully understood; what is more, the deeper we investigate, the greater the number of questions that arise. However, what we truly know is that it must be some kind of director who coordinates all these actors to ensure the right order of each process: the Proliferating Cell-Nuclear Antigen or PCNA.

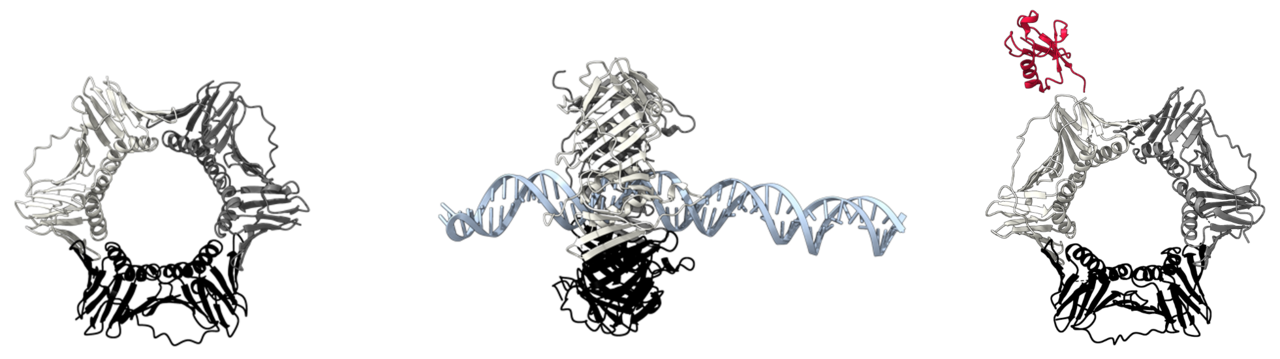
PCNA is a highly conserved homotrimeric ring-shaped protein that encircles DNA and functions as a sliding interaction platform for a wealth of proteins involved in every step required for DNA replication or repair [3]. Thus, PCNA is responsible for recruiting DNA polymerases to accomplish DNA synthesis, DNA repair machinery to address and repair detected DNA damage, alternative polymerases to bypass bulky lesions on DNA, and so on. Now, the most obvious question is how can PCNA recruit the correct enzyme at the right time?
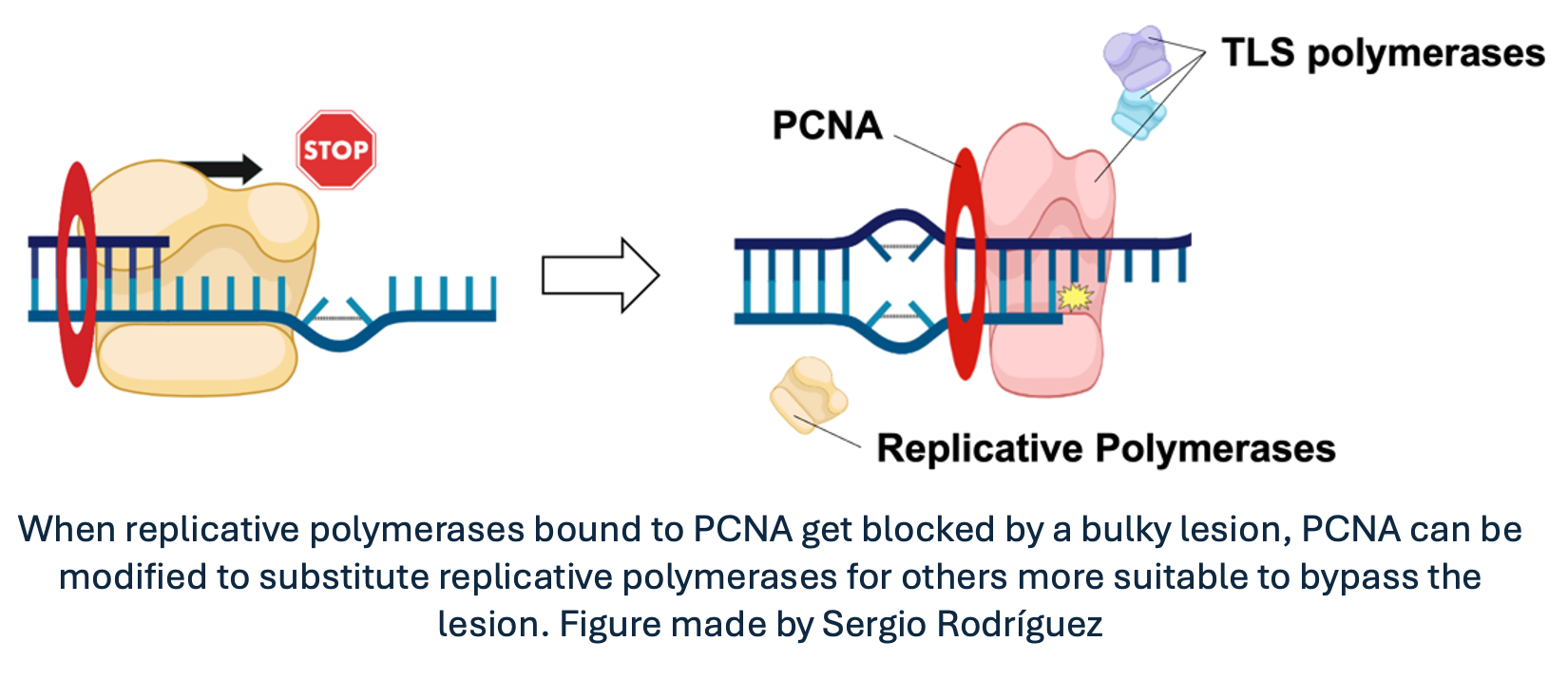
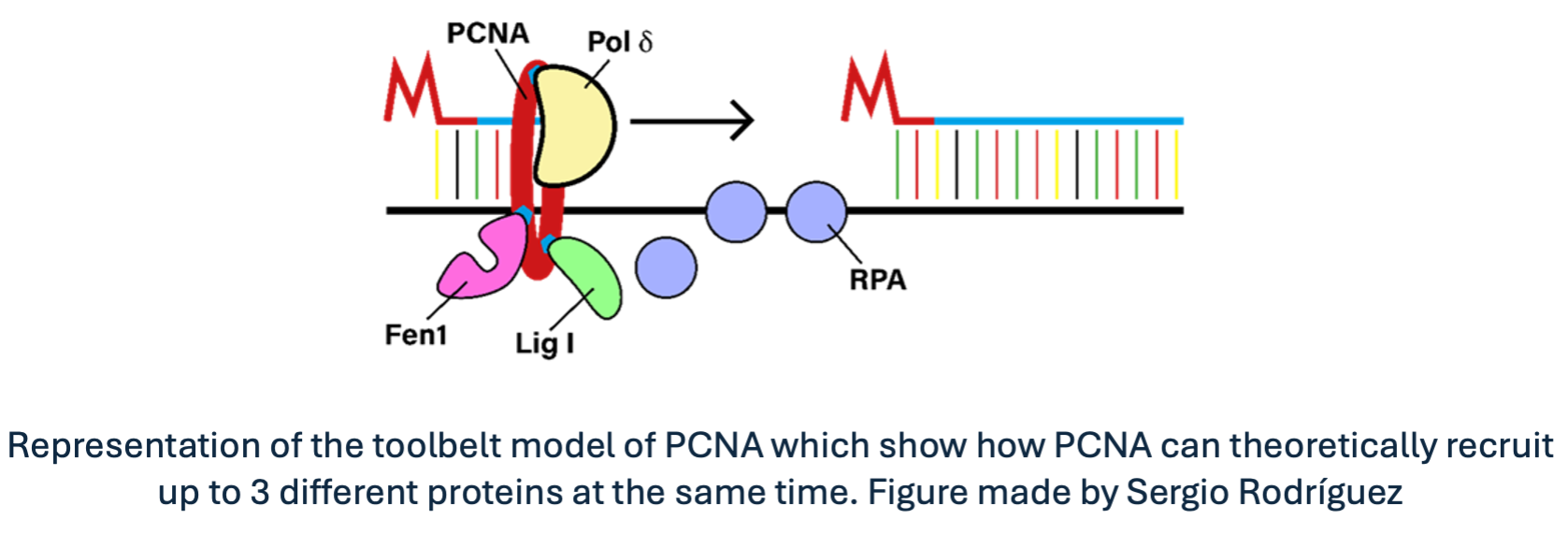
For instance, when encountering DNA lesions, replicative DNA polymerases bound to PCNA get stalled, resulting in potentially harmful effects on cell viability. To avoid this outcome, PCNA can be modified by other proteins to change the affinity for the replicative polymerase. Hence, replicative polymerases are substituted by other types of polymerases that are structurally more suitable for bypassing the lesion and continuing replication coordinately. Furthermore, since PCNA is a homotrimeric protein, it could theoretically bind three different proteins at the same time, acting as a moving toolbelt that places the required tool where and when is required. Awesome, right?
On balance, PCNA is essentially a sliding clamp through the DNA with no enzymatic function; it is just a moving recruitment platform, and it is as easy as pie. However, despite its simplicity, it is the principal director that orchestrates such complex biological processes that keep us alive. Do you find this antithesis now appealing?
FOOTNOTES:
[1] Çengel YA. Eighteen distinctive characteristics of life. Heliyon. 2023 Feb 10;9(3):e13603. doi: 10.1016/j.heliyon.2023.e13603. PMID: 37101483; PMCID: PMC10123176
[2] To know more about the budding yeast S. cerevisiae as a biological model: Wong ED, Miyasato SR, Aleksander S, Karra K, Nash RS, Skrzypek MS, Weng S, Engel SR, Cherry JM (2023) Saccharomyces genome database update: server architecture, pan-genome nomenclature, and external resources. Genetics. 2023 May 4;224(1):iyac191. doi: 10.1093/genetics/iyac191. PMID:36607068
[3] For a deeper understanding of the importance of PCNA: Moldovan, G. L., Pfander, B., & Jentsch, S. (2007). PCNA, the Maestro of the Replication Fork. Cell, 129(4), 665–679. https://doi.org/10.1016/j.cell.2007.05.003
[4] If you want to know more about our current research, I encourage you to read our most recent published work: Zamarreño, J., Muñoz, S., Alonso-Rodríguez, E. et al. Timely lagging strand maturation relies on Ubp10 deubiquitylase-mediated PCNA dissociation from replicating chromatin. Nat Commun 15, 8183 (2024). https://doi.org/10.1038/s41467-024-52542-9
Images by Sergio Rodríguez.





Join the FEBS Network today
Joining the FEBS Network’s molecular life sciences community enables you to access special content on the site, present your profile, 'follow' contributors, 'comment' on and 'like' content, post your own content, and set up a tailored email digest for updates.